By Alexandra Demetriou
An interdisciplinary team of researchers at the University of Southern California has developed a precision drug delivery tool to selectively treat areas of the brain damaged during a traumatic brain injury (TBI). The researchers anticipate that their rapidly deployable intervention could potentially help prevent long-term brain damage in the millions of Americans who sustain a TBI each year.
This invention has been tested using two different therapeutics, and the results appear in the journals Frontiers in Chemistry and Molecular Pharmaceutics.
Stopping the damage before it’s too late
The effects of TBIs on the American populace are nothing short of staggering. TBIs annually cause more deaths and lifelong disabilities than HIV/AIDS, breast cancer, multiple sclerosis and spinal cord injuries combined. Approximately one third of patients die due to secondary complications related to their TBIs, and in 2014, an average of 155 Americans died following a TBI each day. To compound this dramatic human toll, the economic burden of treating TBIs and their long-term side effects is estimated to be $60-76.5 billion each year.
The causes of TBIs range from falls and forceful sports collisions to car accidents and severe blows to the head. Side effects vary widely, from cognitive challenges and dizziness to emotional changes and depression that can endure for days, weeks, or even years after the initial trauma. If effective treatments are not rapidly deployed, a TBI can trigger biochemical changes and inflammatory responses that progressively worsen a patient’s brain damage. To prevent serious side effects, doctors recommend that patients receive treatment within the “golden hour” after sustaining the injury.
Despite the pressing need for effective and rapidly deployable therapies, there are currently no FDA-approved treatments specifically designed for TBIs. Some existing treatments rely on locating the injury within the skull and assessing the damage before proceeding, but this approach can rob precious time from clinicians attempting to stave off permanent brain damage.
A nanoscale solution to a widespread problem
An ideal treatment approach for TBIs would involve a fast-acting, safe, transportable and easily administered drug that could permeate the brain’s protective barrier at the site of damage. A team of USC scientists, led by researchers at the USC Dr. Allen and Charlotte Ginsburg Institute for Biomedical Therapeutics, has mobilized to meet this urgent clinical need: they recently developed a novel drug delivery tool designed to safely and rapidly treat regions of the brain damaged during a TBI.
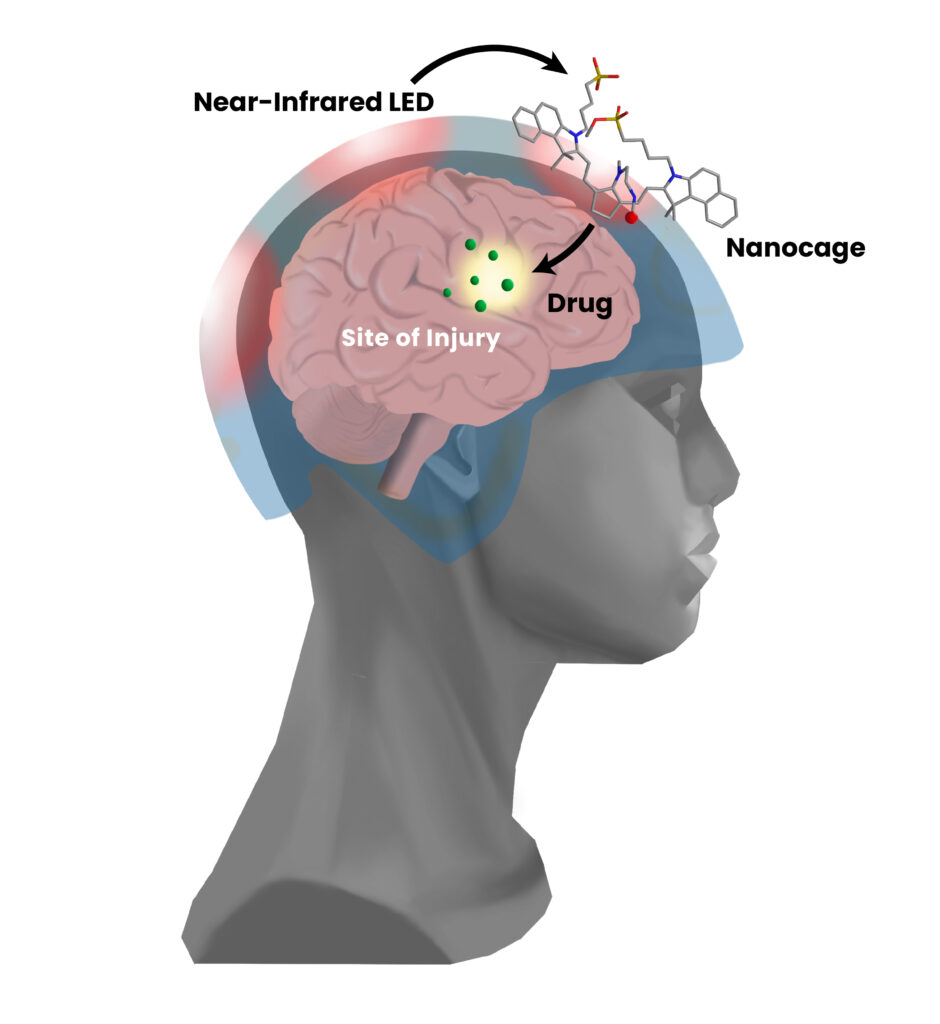
The tool itself is known as a nanocage. As the name suggests, this nanoscale drug carrier works by caging therapeutic agents—in this case, drugs such as gabapentin and cyclosporine A—to carry them within the body. Once a caged drug has migrated through a patient’s circulatory system and made its way to the brain, doctors can beam near-infrared (NIR) light through the patient’s skull to energize the molecule and “open” the cage.
USC scientists have developed an experimental precision treatment for traumatic brain injury (TBI) that involves trapping therapeutic drugs in nanocage carriers before administering treatment. Near-infrared (NIR) light can safely penetrate the skull, and it can be used to “open” the nanocage to release drugs at the site of brain injury. (Image credit: Emily Louie)
The beauty of this approach is multifold. Caging the drug before releasing it at the site of injury minimizes the side effects that one would expect from delivering an active drug throughout the entire body. Moreover, because the brain’s barrier is weakest where it experienced the most trauma, higher concentrations of the drug will enter the brain precisely at the site of damage—thus circumventing the need for time-consuming imaging to locate the damage before proceeding with first-line treatment.
The researchers chose NIR frequency specifically for its ability to safely permeate human tissue without residual side effects, and they designed the nanocage itself to be biodegradable and nontoxic. This emerging intervention is still in its preclinical stages, but the researchers anticipate that it can be packaged as a portable intervention for first responders to immediately administer within a patient’s “golden hour.”
“I’m very hopeful for the impact this drug delivery tool could ultimately have on the millions of patients who experience TBIs each year,” said Mark Humayun, MD, PhD, director of the USC Ginsburg Institute for Biomedical Therapeutics, who is the study’s principal investigator. “A rapid intervention like this could significantly improve patient outcomes, and our team is excited to continue testing this approach to hopefully bring it to the clinic soon.”
Caroline Black, PhD, lead author on the study, says this research was made all the more meaningful because of her personal connection to TBI within her family. “My dad had a severe TBI when he was 17 and I’ve seen how the effects of secondary injury can persist decades after the initial trauma,” said Black, who is a USC-AbbVie postdoctoral fellow specializing in drug delivery sciences at the biopharmaceutical company AbbVie. “Our drug delivery tool has the potential to open new possibilities for rapid treatment of TBI, and I’m excited to see the impact it could have on improving patient outcomes.”
In addition to Humayun and Black, other researchers on the study include Caitlin M. DeAngelo, PhD, of the USC Department of Chemistry; Eugene Zhou and Isaac Asante, PhD, of the USC School of Pharmacy; Stan G. Louie, PharmD, of the USC School of Pharmacy and the USC Dr. Allen and Charlotte Ginsburg Institute for Biomedical Therapeutics; and Nicos A. Petasis, PhD, of the USC Department of Chemistry, the USC School of Pharmacy and the USC Ginsburg Institute.